PART A: Crude Oil Distillation Design
Crude oil is separated into several fractions, each of these fractions are composed of many undefined hydrocarbon components. For simplicity, the crude oil is assumed to be a mixture of five real compounds and will be split into two streams only (top and bottom) using a
distillation column. The components in the crude, amount of feed of each component and their normal boiling
points are shown below.
Components Feed, kmol/hr Boiling Point, Tb, K
1. C7H16 15 371.6
2. C10H22 10 447.3
3. C12H26 25 489.5
4. C15H32 25 543.8
5. C18H38 40 589.5
In the distillate, there can be a maximum of 8% of component 4 (C15H32) and in the bottom,
there can be a maximum of 4% of component 3 (C12H26).
problem 1
find out:
(a) Minimum number of stages required for the separation
(b) Minimum reflux ratio for the separation
(c) Actual number of stages when the actual reflux ration is 1.4 times the minimum reflux ratio.
(d) Actual reflux ratio, when the actual number of stages is 17.
(e) If the actual reflux ratio is 50 times the minimum reflux ratio what happens to the actual number of stages. Comment on your results.
Pure component vapour ( vp P ) pressure (bar) can be find outd using the following Antoine equation:
log10Pvp =A-(B/C+T-273.15) where T is K.
The Antoine constants are shown in the following table for each component.
Components A B C
1. C7H16 4.02023 1263.909 216.432
2. C10H22 4.06853 1495.170 193.858
3. C12H26 4.12285 1639.270 181.840
4. C15H32 4.14849 1789.950 161.380
5. C18H38 4.12710 1894.300 143.300
The Vapour Liquid Equilibrium (VLE) constants ( i k ) can be find outd by Ki=Piv/P where P is the total pressure (1 bar). The relative volatility (αi,r ) of each component (i) with respect to a reference component (r) can be find outd by αi,r= ki/kr
PART B: Crude Oil Hydrotreating
A typical petroleum refinery is a complex chemical processing and manufacturing unit, with crude oil feedstock going in and refined fractions (products). Refining occurs by fractionation (distillation) of crude oil into a series of product streams based on boiling ranges for each fraction. Figure below shows a typical layout for an oil refinery.
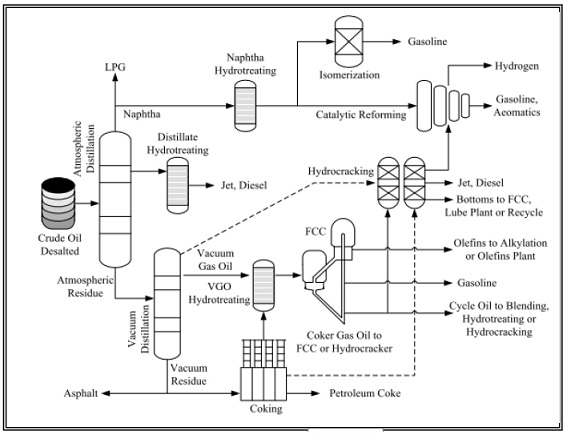
Crude oil is a very complex material consisting of different hydrocarbon compounds in addition to sulfur, nitrogen, oxygen and some metallic compounds, containing nickel, vanadium, iron and copper. The presence of sulfur, nitrogen, oxygen and metallic compounds in crude oil has a significant impact upon the quality of oil products in addition to the harm they can cause.
problem 2:
Study on your own (books, web based materials, journals) and
a) prepare a 100 words summary on the harmful effects of sulfur, nitrogen, oxygen and some metallic compounds, containing nickel, vanadium, iron and copper if they are present in the oil fractions obtained by crude oil distillation
b) prepare a 500 words summary on hytrotreating process and how it is currently carried out in existing refineries Study the references (electronic copies are attached with this coursework) and
c) prepare a 400 words summary on the new method of hydrotreating
While writing on items (b) & (c) reflect on the design & operational issues of the existing hytrotreating and the new hydrotreating processes.
You must list down all the references used. You must not exceed the word limit (penalty marks will apply).
Text in the report should be word-processed, but calculations may be neatly hand-written to save time. If you use spreadsheet calculations, you must describe the calculation method, illustrating your explanation with a sample hand calculation. Diagrams may be either hand- or computer-drawn.