problem 1: Strained alkenes like cyclopropene or norbornene bind unusually strongly to metals. Recommend a reason why.
problem 2: Alkynes readily bridge M-M bonds, in which case they act as 2e donors to each metal. Sketch the product of the reaction below, pointing out the hybridization of the C atoms.
PhCCPh + Co2(CO)8 → (µ2-PhCCPh)Co2(CO)6 + 2CO
problem 3: Draw as many bonding modes for cyclooctatetraene as you can think of.
problem 4: Sketch the three π-MOs of the allyl anion, [C3H5]–. Draw metal d-orbitals which can interact with these MOs and name the type of bonding (e.g. M→L π-acceptor).
problem 5: The M-P distance in (η5-C5H5)Co(PEt3)2 is 221.8 pm and the P-C distance is 184.6 pm. The corresponding distances in [(η5-C5H5)Co(PEt3)2]+ are 223 pm and 182.9 pm. Account for the changes in such distances as the former complex is oxidized.
problem 6: Predict the product of the reactions between:
a) [Ru(η5-C7H9)(η6-C7H8)]+ and H–
b) [W(η5-C5H5)2(η3-C3H5)]+ and H–.
problem 7: Ligands of type X–Y only give 3c-2e “agostic” bonds to transition metals if X = H and Y lacks lone pairs. Why do you think this is so (consider alternative structures if X and Y are not H)?
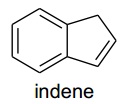
problem 8: [IrH2(H2O)2(PPh3)2]+ reacts with indene to give [Ir(C9H10)(PPh3)2]+
A) On heating, this species rearranges with H2 loss to provide [IrH(C9H7)(PPh3)2]+
B) Only A reacts with ligands like CO to displace C9H7. What do you think are the structures of A and B?