Calculate the heats of combustion for the following reactions from the standard enthalpies of formation listed in Appendix 3.(ΔH°f(HgO(s)) = -90.7kJ/mol and ΔH°f (FeO(s)) =-272 kJ/mol.)(a) 2 Fe(s) +O2(g) -> 2FeO(s)
1 kJ
(b) 2 C6H6(l) + 15O2(g) 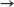 12CO2(g) + 6 H2O(l)
12CO2(g) + 6 H2O(l)
2 kJ